on SANOFI-AVENTIS (EPA:SAN)
Dupixent Approved in the US as First Biologic for COPD Patients
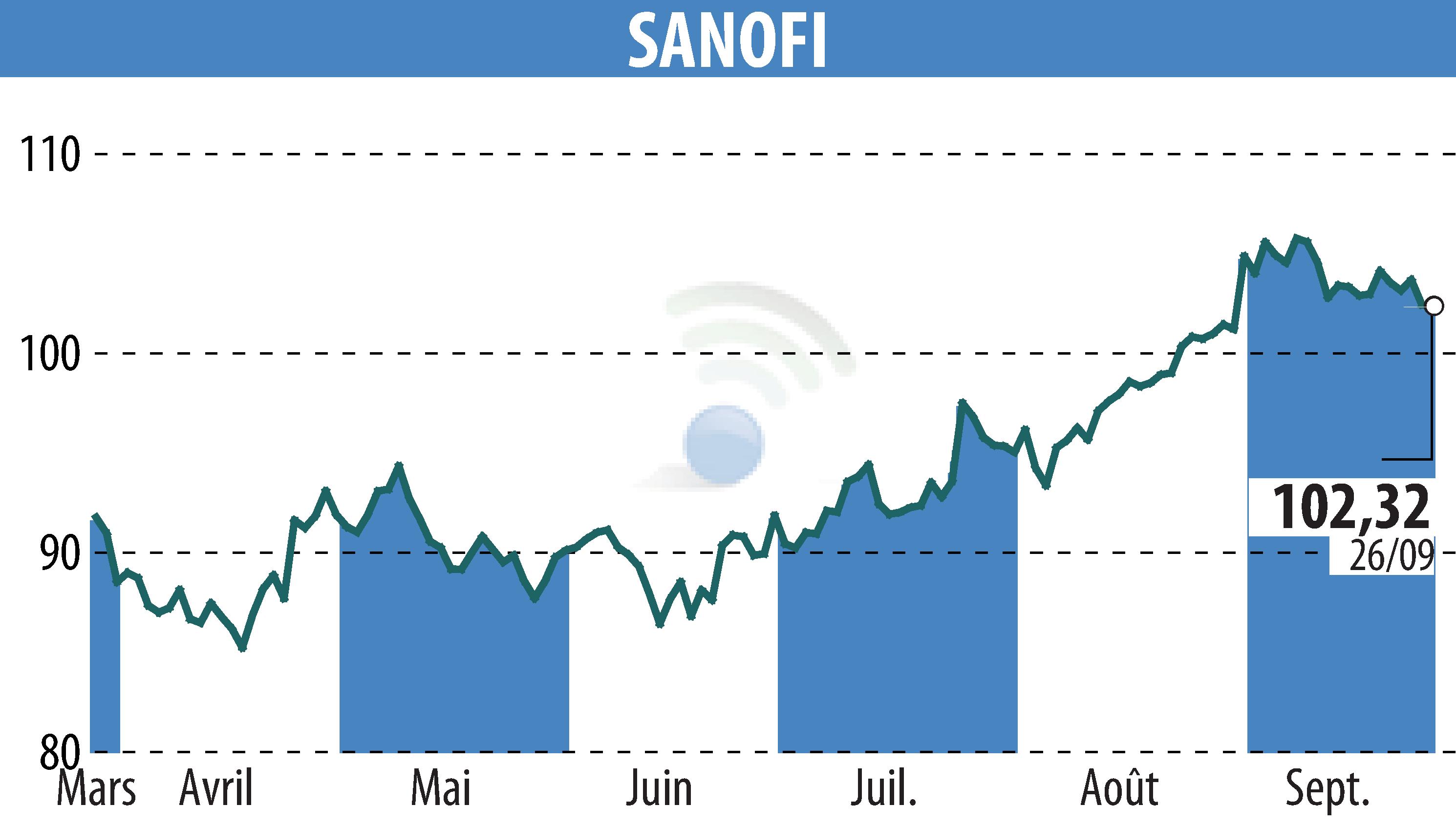
Sanofi-Aventis announced that the FDA has approved Dupixent (dupilumab) as an add-on treatment for adults with inadequately controlled chronic obstructive pulmonary disease (COPD) and an eosinophilic phenotype. This drug is now the first biologic medicine approved in the US to treat this subset of COPD patients.
The approval follows positive results from two phase 3 studies, BOREAS and NOTUS, which demonstrated a 30% and 34% reduction in annualized rates of moderate or severe COPD exacerbations over 52 weeks. Additionally, patients showed improved lung function and a better quality of life when compared to placebo. Safety results were consistent with the known profile of Dupixent.
Before this US approval, Dupixent had already gained approval in the EU and China. The drug is highly regarded by pulmonologists and holds a leading position in new-to-brand prescriptions for various FDA-approved indications.
This advancement offers new hope for approximately 300,000 US adults suffering from inadequately controlled COPD, aiming to provide improved breathing and reduced exacerbations.
R. E.
Copyright © 2024 FinanzWire, all reproduction and representation rights reserved.
Disclaimer: although drawn from the best sources, the information and analyzes disseminated by FinanzWire are provided for informational purposes only and in no way constitute an incentive to take a position on the financial markets.
Click here to consult the press release on which this article is based
See all SANOFI-AVENTIS news
